Antiviral Drug Screening
ANTIVIRAL SCREENING
Antiviral Drug Screening Services
Antiviral testing is done to reveal the antiviral properties of drugs, biomolecules, and other small molecules.
There are several ways to run an antiviral test. For instance, compounds can be tested at a single concentration or at multiple concentrations. When multiple concentrations are tested, it’s often possible to calculate the EC50 value.
Also, the test compound can be added at different stages of virus infection: e.g., pre-infection (before introducing the virus) or post-infection (before introducing the virus). Test compounds might also be pre-incubated with the virus (neutralization). These various tests can help reveal how an antiviral works (e.g., does it neutralize the virus or does it stop viral entry).
In our immunofluorescence assay, it is also possible to stop the assay after a single round of replication (before synthesis of new viral progeny) or after multiple rounds (spreading infection).
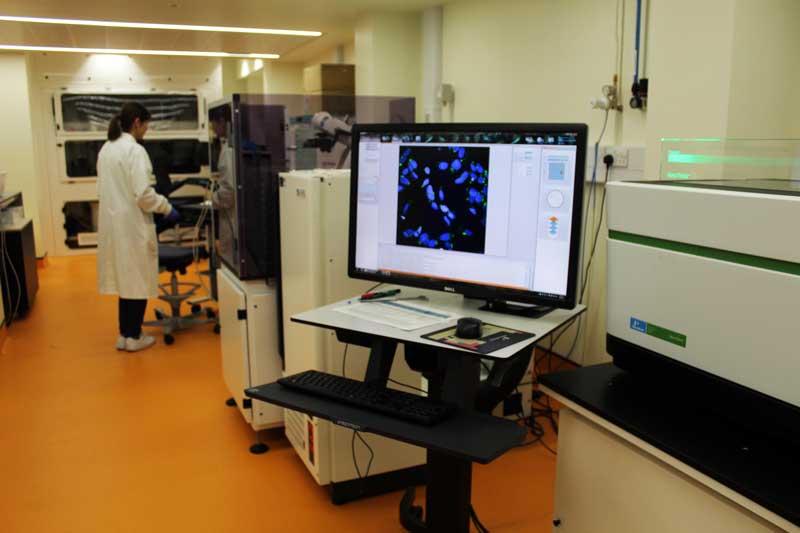
Our Approach
How our antiviral drug screening is done.
High content imaging: At the end of the infection period, cells are fixed and immuno-stained for a viral antigen. Antiviral activity is determined by quantifying the percentage of infected cells. For this, we use a high content imaging instrument (CellInsight CX5, Thermo Fisher Scientific). An Orbitor plate handling robotic system allows for the processing of up to 60 plates.
The amount of virus we use depends on the virus, the host cells, and the incubation time. When only a single round of virus replication is required, a higher virus to cell ratio (multiplicity of infection, or MOI) is needed, while low MOIs are used in spreading infection assays.
Cytotoxicity testing: We also run a cytotoxicity test in parallel. In this test, cells are treated with the same concentrations of the compound but in the absence of virus. Any cytopathic effect of the test compound will be considered when interpreting the antiviral assay. If the compounds are tested in a dose-response manner, a TC50 value will also be generated.
Testing in triplicate with controls: Our antiviral assays are performed in triplicate, and they include infected and uninfected controls. Whenever available, a positive control (a compound known to inhibit viral infection in the assay system) is also included.
Drug screening services are available for the following viruses:
Cytomegalovirus (CMV)
Dengue virus (serotypes 1-4)
Human coronavirus NL63 (HCoV-NL63)
Human coronavirus 229E (HCoV-229E)
Human immunodeficiency virus (HIV)
Influenza (A and B)
Respiratory syncytial virus (RSV)
Ross River virus
SARS-CoV-2
Semliki Forest virus
Sindbis virus
Vaccinia virus
Zika virus (African and Asian)
Other viruses may be available on request.
Subscribe

